|
关注:1
2013-05-23 12:21
求翻译:The values of ∆H f and S° in Tables 1-3 can be used to calculate the gas-phase ∆G f ° at 298.15 K. This can be done by calculating the entropy of formation, ∆S f °, found by subtracting off the entropies of the atomic standard states (Table 2) from the virtual entropy of the compounds. For example, ∆S f °(CH2Cl-CH2-CH2是什么意思?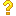 待解决 待解决
 悬赏分:1
- 离问题结束还有 悬赏分:1
- 离问题结束还有
The values of ∆H f and S° in Tables 1-3 can be used to calculate the gas-phase ∆G f ° at 298.15 K. This can be done by calculating the entropy of formation, ∆S f °, found by subtracting off the entropies of the atomic standard states (Table 2) from the virtual entropy of the compounds. For example, ∆S f °(CH2Cl-CH2-CH2
问题补充: |
|
2013-05-23 12:21:38
ΔH F和S °的表1-3中的值可以被用来计算气相ΔG ˚F ° 298.15 K。这可以通过计算地层的熵来完成,△S ˚F °,减去发现
|
|
2013-05-23 12:23:18
∆H f和S°的价值在表1-3可以用于计算气相∆G f °在298.15 K。这可以由计算熵完成形成, ∆S f °,找到通过减去原子标准状态(从化合物的真正熵的表2)的熵。例如, ∆S f ° (CH2Cl-CH2-CH2Cl)和∆G f ° (CH2Cl-CH2-CH2Cl)从以下表示被计算:
|
|
2013-05-23 12:24:58
正在翻译,请等待...
|
|
2013-05-23 12:26:38
正在翻译,请等待...
|
|
2013-05-23 12:28:18
正在翻译,请等待...
|
湖北省互联网违法和不良信息举报平台 | 网上有害信息举报专区 | 电信诈骗举报专区 | 涉历史虚无主义有害信息举报专区 | 涉企侵权举报专区